Shantou, May 7, 2026 – Dr. Kangli Dong from the Department of Biomedical Engineering, College of Engineering, Shantou University, has published a research paper titled "Optimal stochastic tracking control for brain network dynamics" as the first author in Communications Biology, a prestigious journal under the Nature Portfolio (ranked in the first quartile by CAS). This study proposes a novel computational framework that shifts the paradigm from "reaching a target state" to "tracking target dynamics," offering new insights for personalized and low-energy brain stimulation interventions.
Network Control Theory (NCT) is widely used to interpret brain stimulation effects and optimize non-invasive techniques such as Transcranial Magnetic Stimulation (TMS) and Transcranial Direct Current Stimulation (tDCS). However, traditional optimal control frameworks typically aim to transfer the brain to a specific target state at a single time point. This approach fails to fully capture the continuous evolution and stochastic noise inherent in real brain activity.
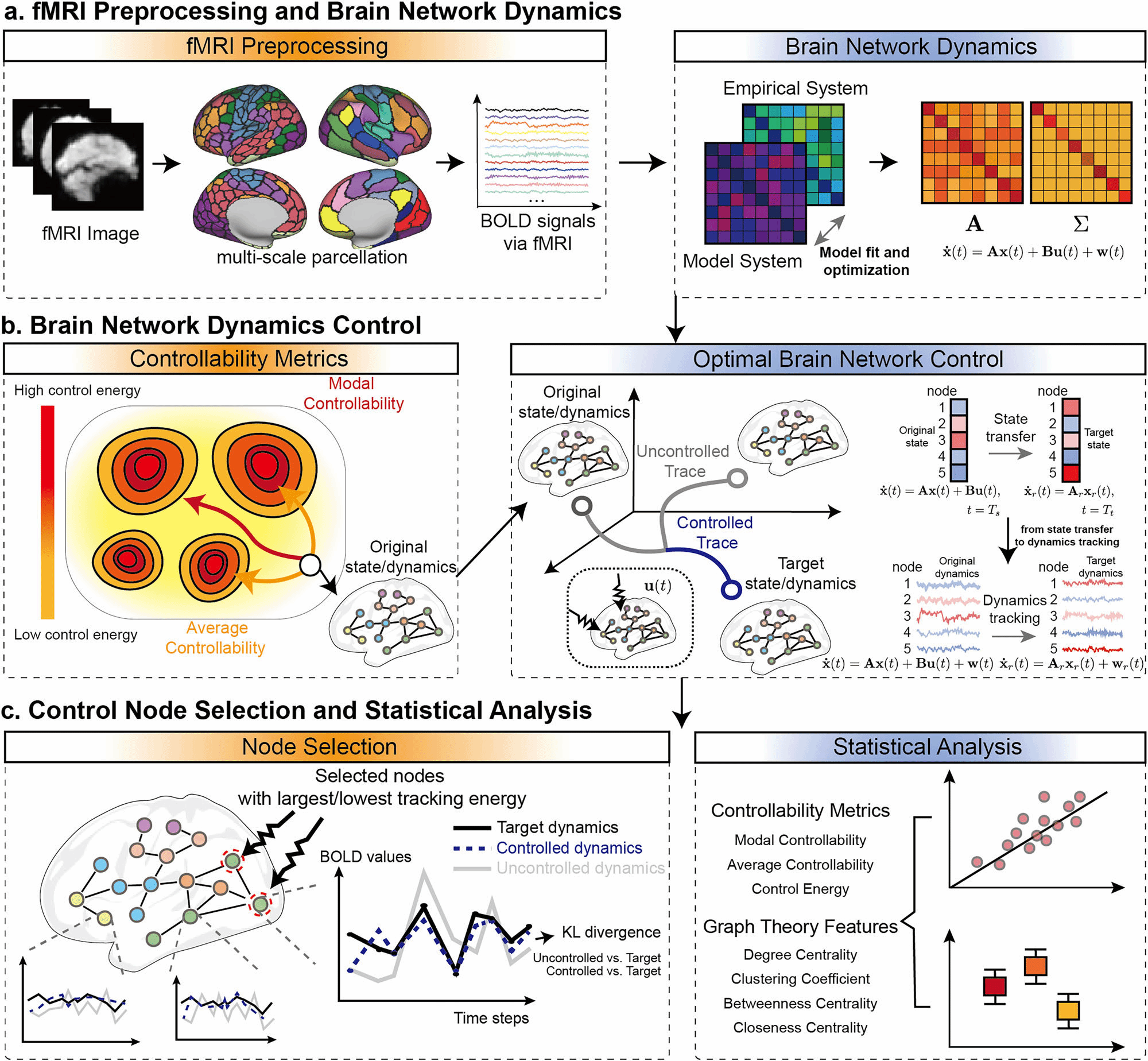
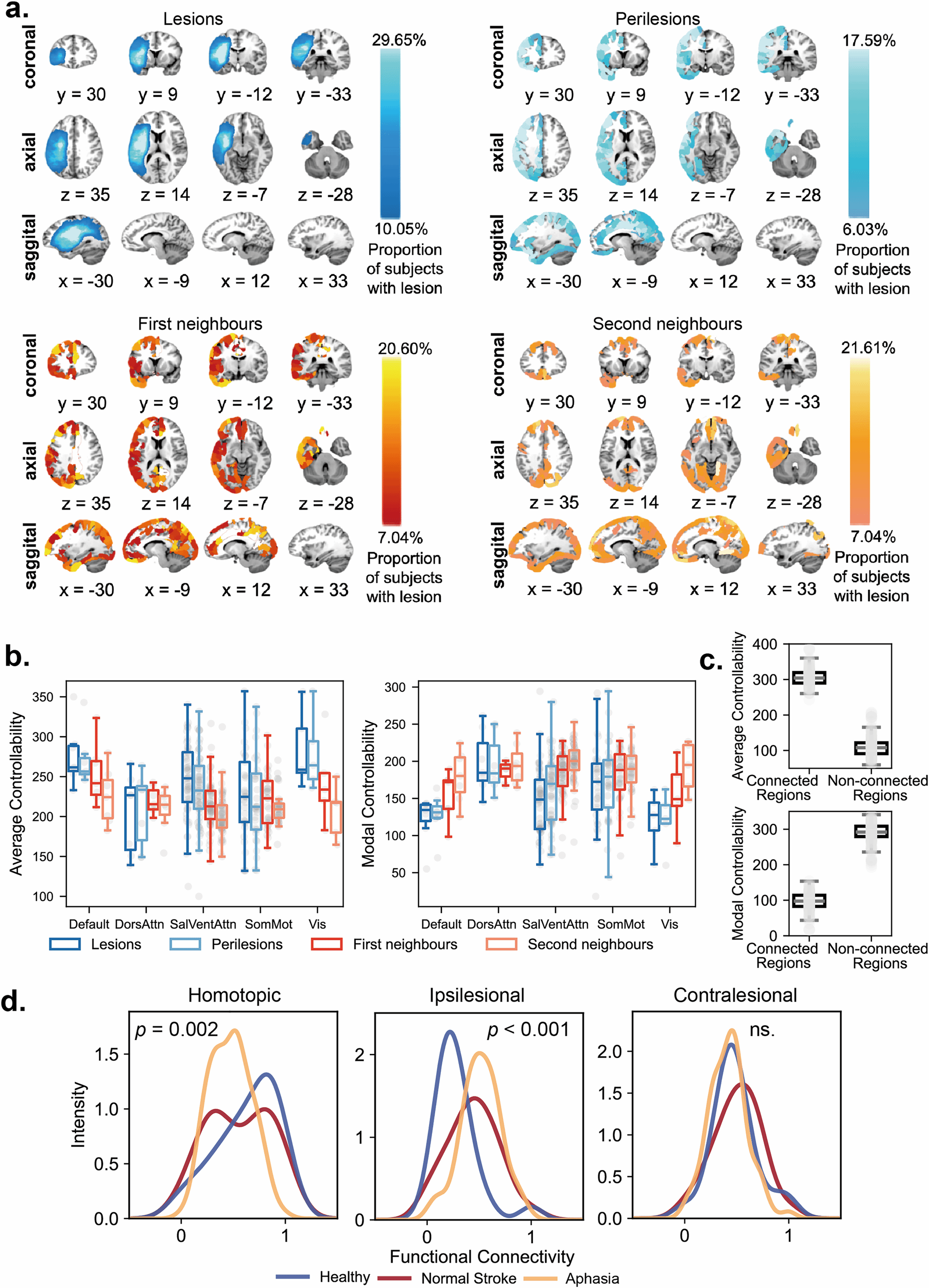
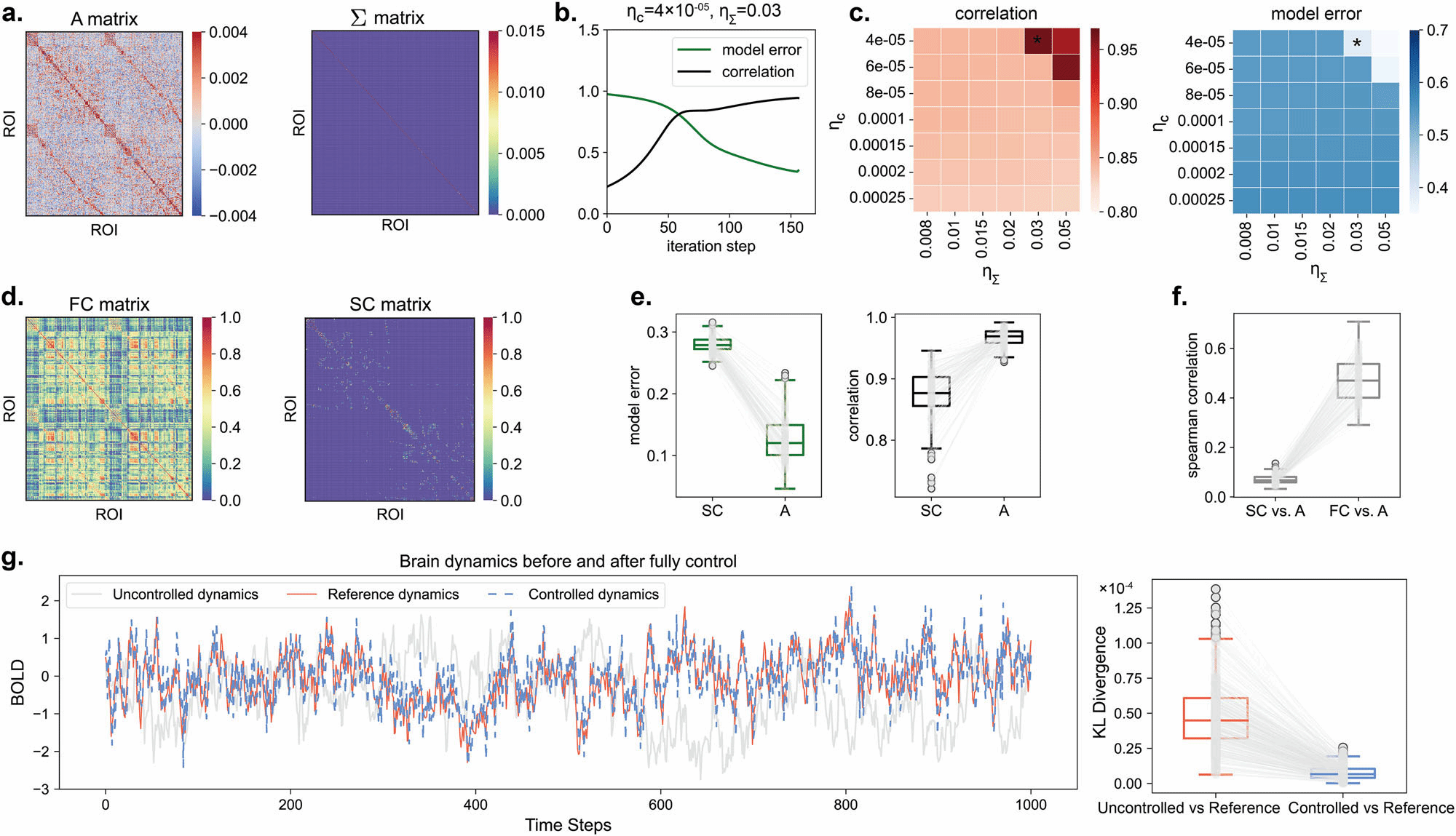
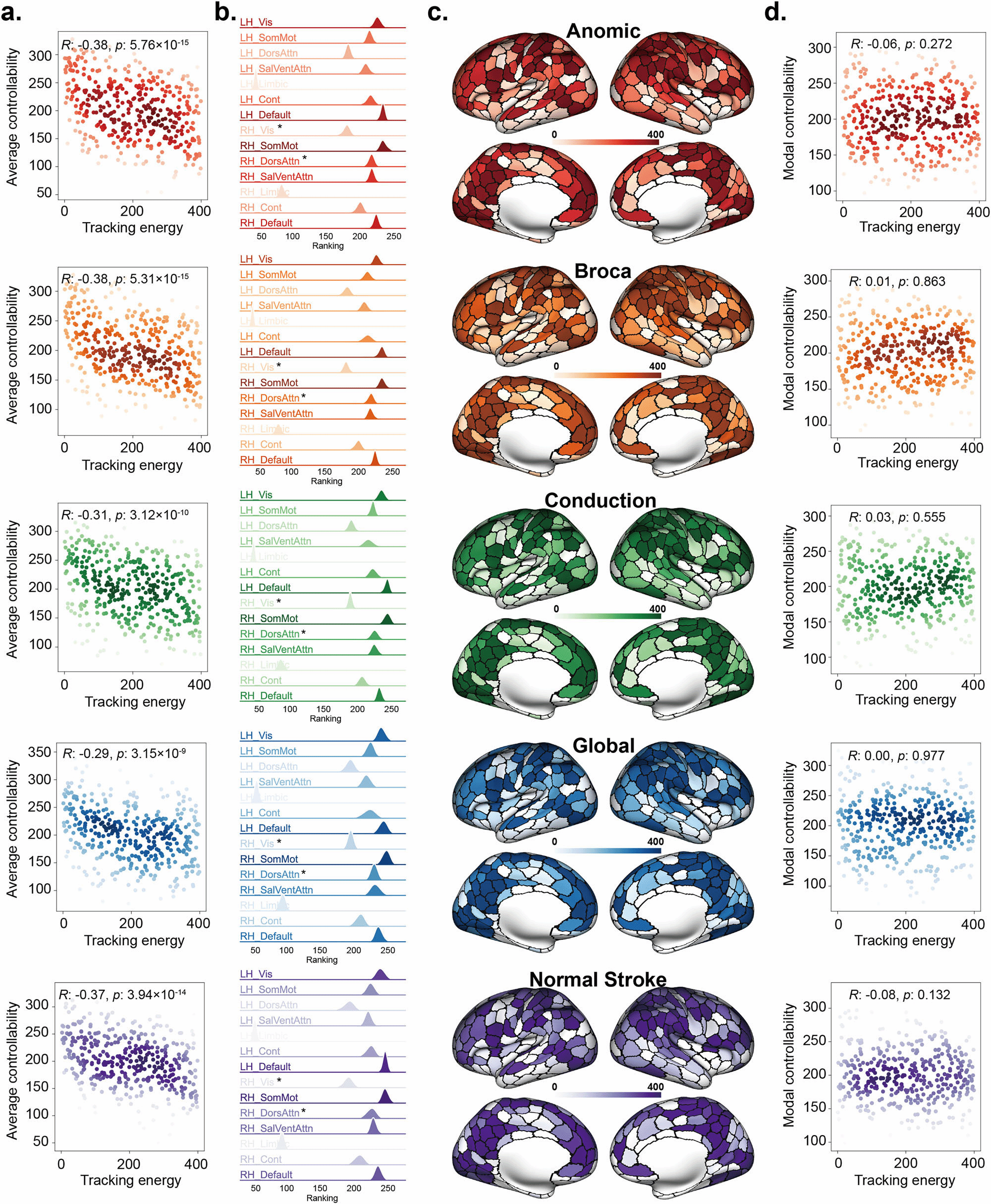
To address this limitation, Dr. Dong's team proposed an Optimal Stochastic Tracking Control framework. Instead of merely pushing the brain to a static state, this method aims to synchronize pathological brain dynamics with healthy target dynamics in terms of statistical properties. Using fMRI and DWI data from stroke and post-stroke aphasia patients, the team employed gradient descent optimization to estimate the coupled matrix and variance matrix of the brain network system.
The study yielded significant findings:
Theoretical Advancement: The research successfully advanced brain network control from "optimal state transfer" to "optimal dynamic tracking," making the model more consistent with the actual goal of long-term rehabilitation.
Key Correlation: The tracking energy was found to be significantly negatively correlated with the brain network's average controllability—a relationship not observed in traditional state transfer control.
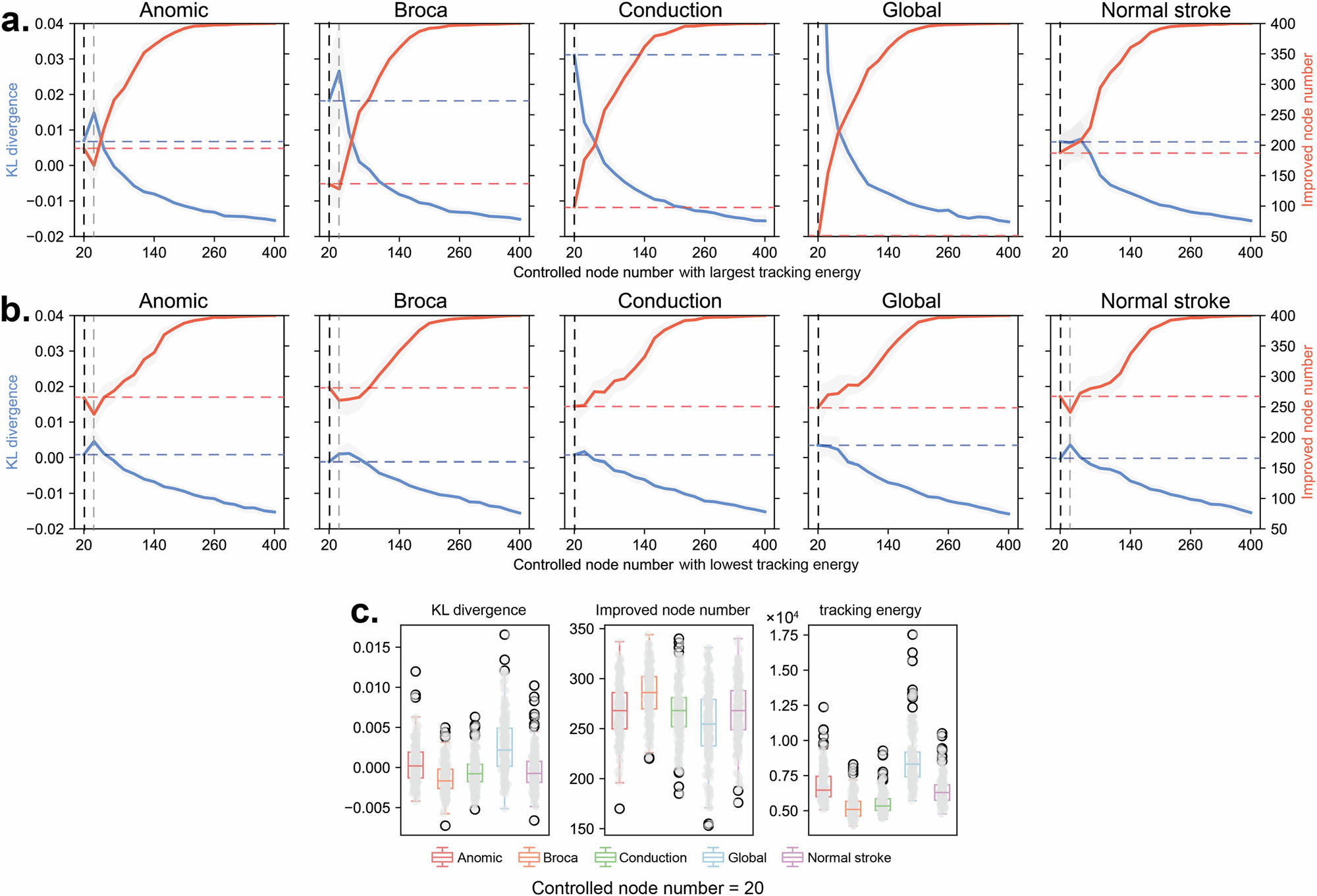
High Efficiency: In a 100-dimensional brain network system, controlling just 5 nodes with the lowest tracking energy improved the dynamics of over 90% of the nodes.
This discovery suggests that the optimal stochastic tracking control framework can identify potential stimulation targets with lower costs and higher efficiency. It provides a robust theoretical foundation for guiding brain stimulation interventions in neurological disorders such as stroke and aphasia.
Looking ahead, this framework offers a new computational tool for clinical applications. It can evaluate the theoretical control cost of different brain regions as stimulation targets and help explore strategies to achieve maximum dynamic improvement with minimal intervention. By integrating brain dynamic models, analysis of language-related cortical regions, and multimodal neuroimaging data, this method holds great promise for advancing precision brain stimulation and personalized neurorehabilitation.
DOI: https://doi.org/10.1038/s42003-025-09184-4
Text: College of Engineering